










- Stock: In Stock
- Product code: 00-00000266
- Weight Brutto: 1.50kg
- SKU: RS-F2-BMAM-01
Formlabs BioMed Amber Resin Cartridge 1 L
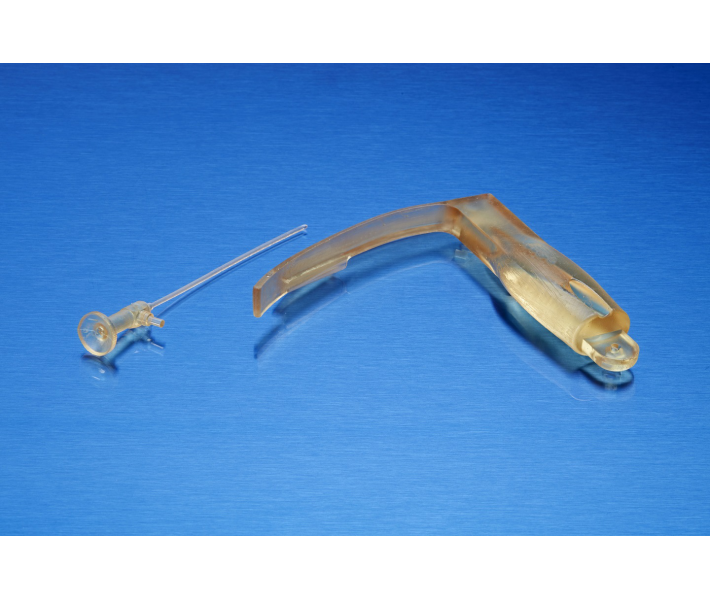
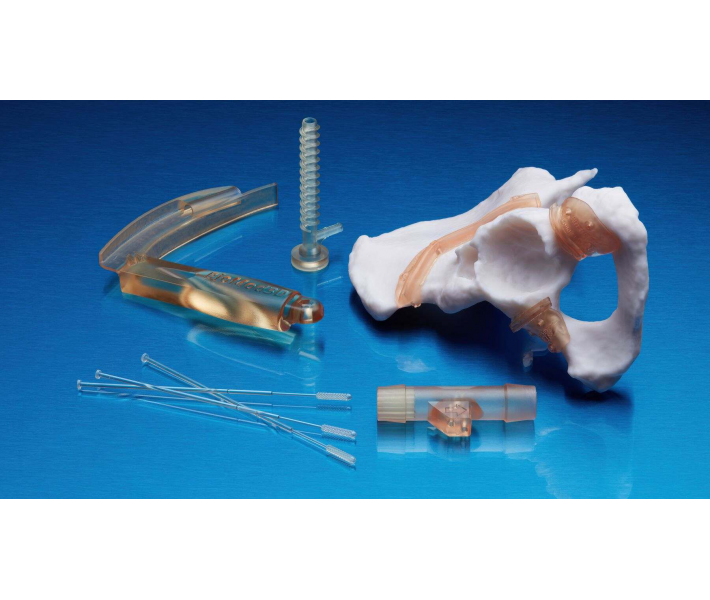
The BioMed Amber technical resin is a rigid material designed for the production of biocompatible parts via 3D printing. It stands out for its high dimensional accuracy, rigidity, and durability, making it ideal for applications requiring brief contact with skin or mucous membranes.
This material has been developed specifically for the Formlabs printer ecosystem. It is manufactured in cleanrooms within Formlabs facilities, which hold ISO 13485 certification. Furthermore, finished parts are fully compatible with standard solvent-based disinfection and sterilisation methods.

Thanks to its robustness and reliability, BioMed Amber Resin is the ideal choice for 3D printing in the following sectors:
- Manufacture of medical devices and their components.
- Surgical guides and drilling templates for implants.
- Customised surgical planning tools.
- Drug delivery devices.
- Bioprocessing equipment.
- Research and development (R&D) in the healthcare sector.

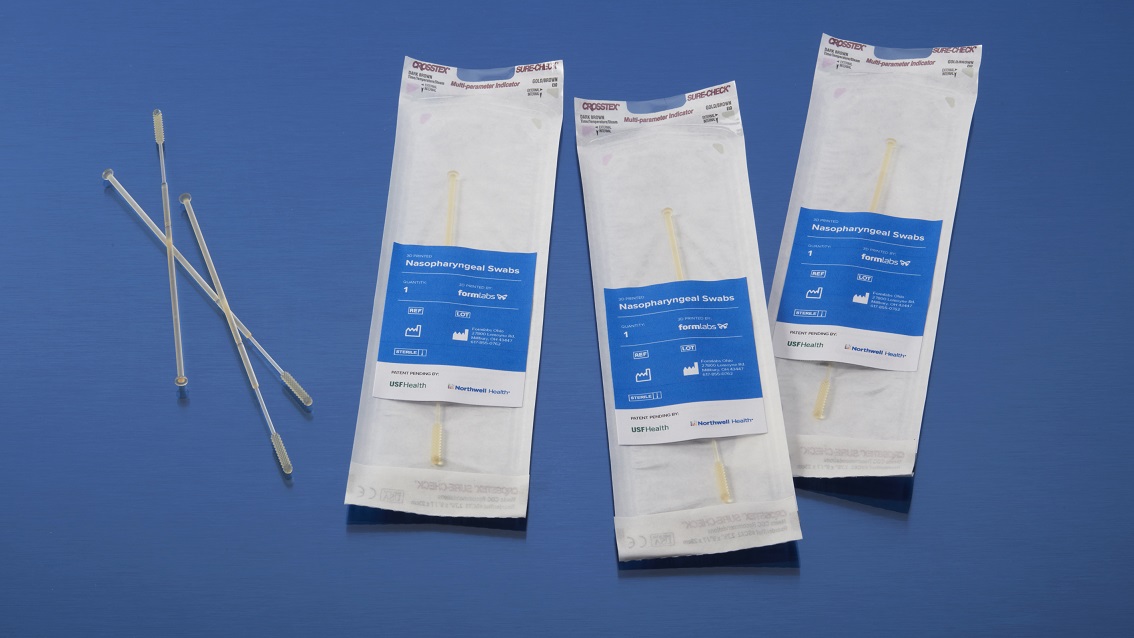
The Formlabs BioMed resin range offers medical-grade materials that comply with ISO 13485 standards. These solutions are certified for use cases where the part must come into direct contact with the patient, ensuring the highest level of biological safety.
Mechanical Properties of BioMed Amber Resin:
| Mechanical Property | After Curing | Test Method |
|---|---|---|
| Tensile Properties | ||
| Tensile Strength | 73 MPa | ASTM D638-10 (Type IV) |
| Young's Modulus | 2.9 GPa | ASTM D638-10 (Type IV) |
| Elongation | 12% | ASTM D638-10 (Type IV) |
| Flexural Properties | ||
| Flexural Strength | 103 MPa | ASTM D790-15 (Method B) |
| Flexural Modulus | 2.5 GPa | ASTM D790-15 (Method B) |
| Disinfection and Sterilisation | 70% Isopropyl Alcohol (5 min) / Autoclave (134 °C – 20 min) | |
BioMed Amber resin meets the most stringent standards for biocompatibility risk:
| ISO Standard | Test Description |
|---|---|
| EN ISO 10993-5:2009 | Non-cytotoxic |
| ISO 10993-10:2010 | Non-irritating / Non-sensitising |
1 Material properties may vary depending on part geometry and slicer settings.
2 Data obtained after post-processing in Form Wash and Form Cure (60 °C for 30 min).
3 Resin tested at NAMSA World (Ohio, USA).




























































