









- Stock: In Stock
- Product code: 00-00012996
- Weight Brutto: 1.50kg
- SKU: RS-F2-BMDU-01
BioMed Durable Resin: Strength and Medical-Grade Performance for Your Prints
The BioMed Durable Resin is a transparent photopolymer designed specifically for the medical sector, ideal for applications where resistance to impact, fracture, and abrasion is critical. This USP Class VI material is suited to a wide range of healthcare uses requiring prolonged skin contact (over 30 days) and mucosal contact (over 30 hours), or brief contact with tissue, bone, and dentine (under 24 hours).
BioMed Durable is manufactured at FDA-registered, ISO 13485-certified Formlabs facilities. It also holds USP Class VI certification, making it a safe and reliable choice for pharmaceutical applications and drug delivery systems.
Why Choose BioMed Durable Resin?
- Guaranteed biocompatibility: Approved for prolonged contact with skin and mucous membranes, as well as short-term contact with internal tissue, bone, and dentine.
- Medical-grade certification: Manufactured under strict quality controls (ISO 13485) at FDA-registered facilities and certified to USP Class VI.
- Superior strength and durability: Printed parts withstand impacts of up to 98 J/m, resisting chipping or fracturing under knocks and drops.
- Crystal-clear transparency: Delivers excellent clarity, enabling the creation of functional parts that are both aesthetically appealing and allow internal visualisation.
- Easy post-processing: Achieve smooth surfaces and professional finishes with straightforward post-print processing.
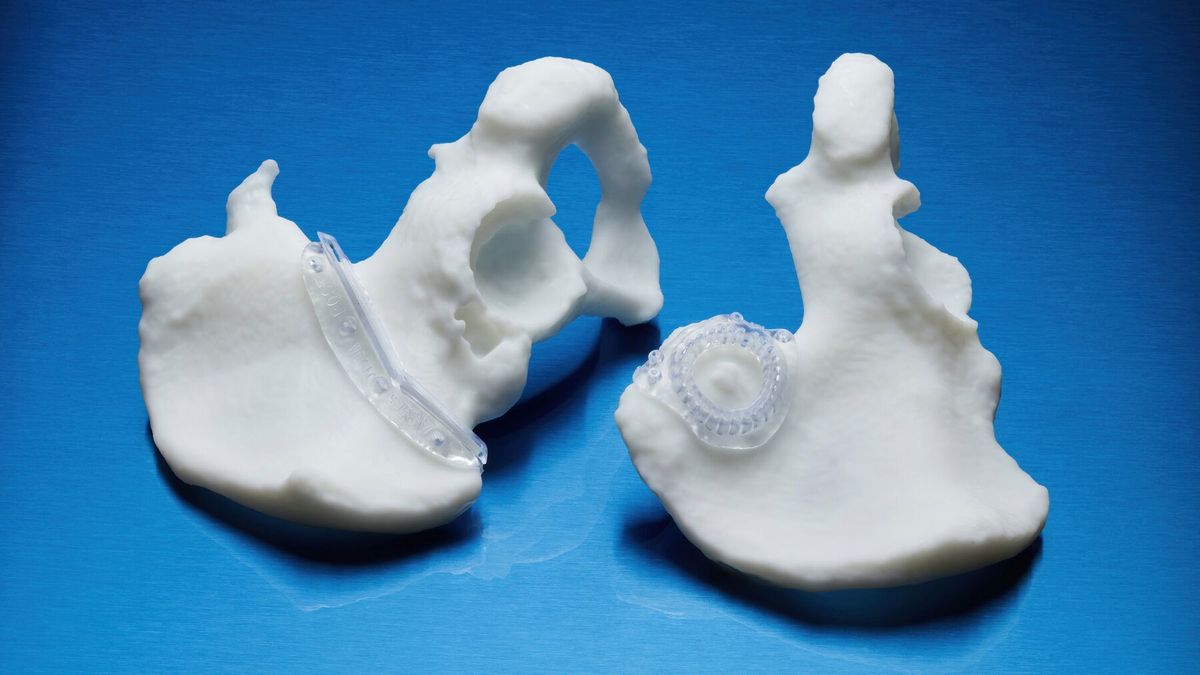
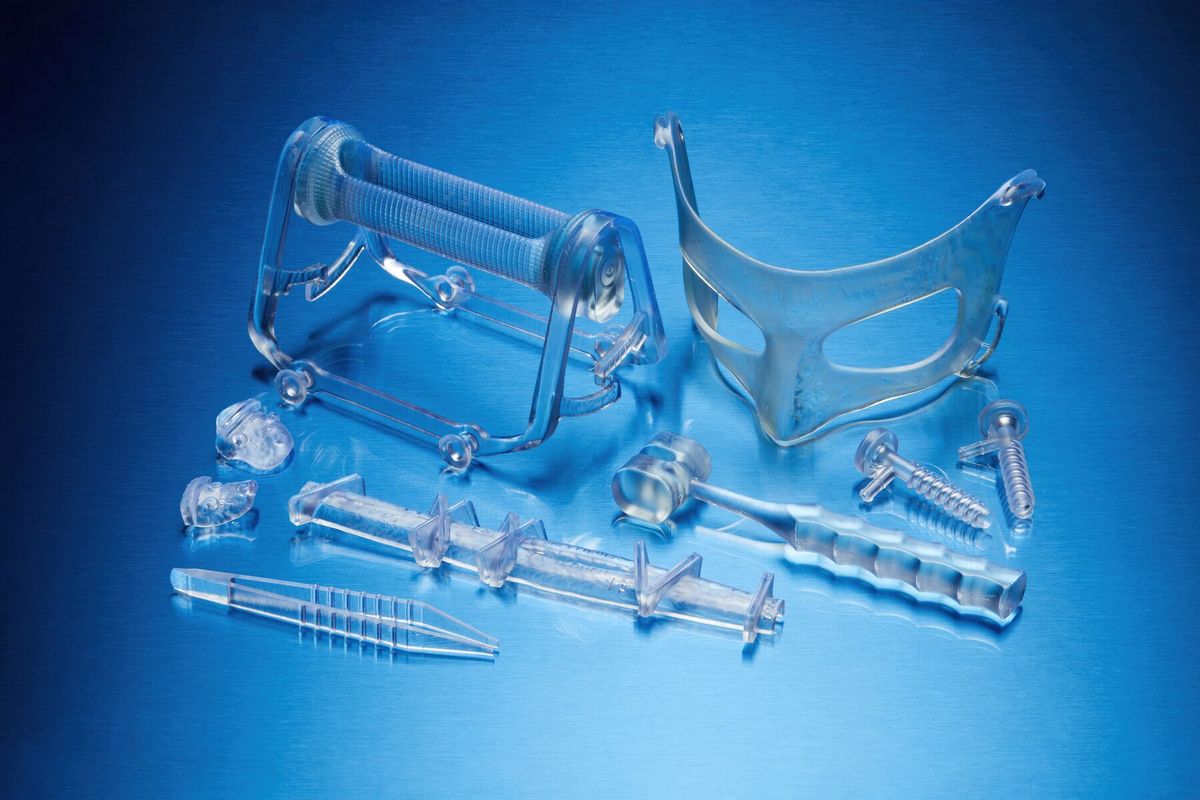
Key Applications of BioMed Durable Resin*
- Patient-specific instruments (PSI): Create innovative guides and instruments tailored to each patient's anatomy, ready for use at the point of care.
- Custom tooling: Ideal for producing end-use jigs and components that require biocompatibility and high mechanical strength.
- Surgical instrumentation: Print surgical instruments, fixations, and single-use tools with complete confidence.
Technical Specifications
| Printing technology | SLA |
| Post-curing | Required |
| Biocompatibility | Yes |
| Tensile strength | 29.1 MPa |
| Tensile modulus | 994 MPa |
| Elongation at break | 33% |
| Flexural strength | 21 MPa |
| Flexural modulus | 643 MPa |
| Shore D hardness | 75D |
| Notched Izod impact | 98 J/m |
| Heat deflection temperature (0.45 MPa) | 46 °C |
| Layer height (microns) | Form 3B, Form 3B+, Form 3BL – 100 |
| Printer compatibility | Form 3B, Form 3B+, Form 3BL |
| Resin tank compatibility | Form 3 RT V1/V2/V2.1, Form 3L RT V1/V2 |
| Build platform compatibility | Form 3 BP, Form 3 BP 2, Form 3 Stainless Steel BP, Form 3L BP |
| Volume (L) | 1 |
| Country of manufacture | USA |
If you have any questions about the compatibility or applications of this material, please get in touch with our technical support team via the EXPERT3D contact page.
Downloads and Documentation:
Technical Data Sheet (TDS), PDF (EN)
Safety Data Sheet (SDS), PDF (EN)




























































