












- Stock: In Stock
- Product code: 00-00001641
- Weight Brutto: 1.50kg
- SKU: RS-F2-BMCL-01
Formlabs BioMed Clear Resin Cartridge 1L
The BioMed Clear Resin is a high-performance photopolymer with USP Class VI certification, formulated from an advanced blend of methacrylic acid esters and photoinitiators. Designed for professionals who demand the very best.
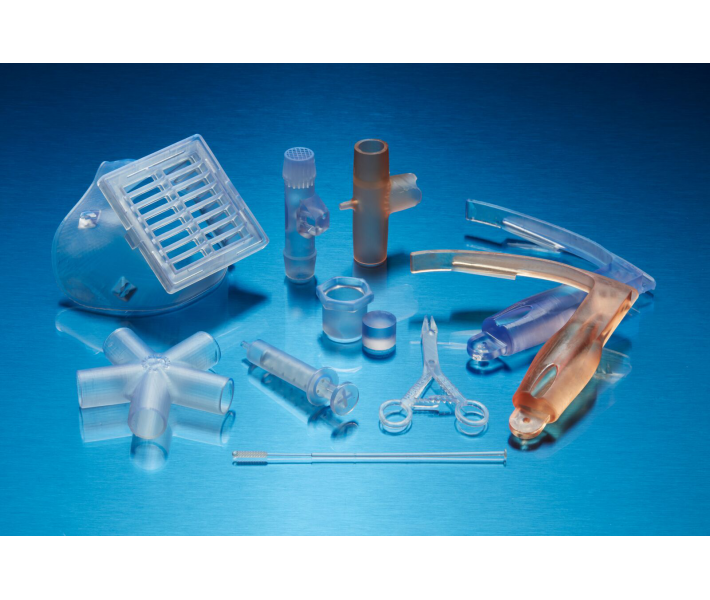
This technical resin cures via light and is specifically designed for the Additive Manufacturing of biocompatible, transparent, and rigid medical parts suitable for prolonged bodily contact (more than 30 days). It is the ideal material for producing medical devices and components that require water resistance, high impact toughness, and compatibility with standard sterilisation methods.

BioMed Clear Resin stands out as a hard, robust material — perfect for applications requiring direct contact with skin or mucous membranes. This material is recommended when you need professional results with low water absorption and high wear resistance in sectors such as:
- Manufacturing of medical devices and their components.
- Surgical planning guides and implant sizing.
- Drug delivery devices.
- Bioprocessing equipment.
- Research and Development (R&D) in biomedical engineering.
Workflow note: To ensure the strict biocompatibility of BioMed Clear, you must use accessories dedicated exclusively to this material. You will need a resin tank, a build platform, and a Form Wash that have not been in contact with any other resins.
At EXPERT3D, we share Formlabs' vision: bringing the most advanced tools to every medical facility to improve patient care. BioMed resins comply with ISO 13485 standards, being medical-grade materials developed for healthcare professionals worldwide. Trust this material for real clinical applications.
Technical Data Sheet: BioMed Clear Resin Properties
| Mechanical Properties | Post-cured1,2 | Method |
| Tensile Strength | 52 MPa | ASTM D638-10 (Type IV) |
| Young's Modulus | 2080 MPa | ASTM D638-10 (Type IV) |
| Elongation at Break | 12% | ASTM D638-10 (Type IV) |
| Flexural Strength (at 5% strain) | 84 MPa | ASTM D790-15 (Method B) |
| Flexural Modulus | 2300 MPa | ASTM D790-15 (Method B) |
| Shore D Hardness | 78 D | ASTM D2240-15 (Type D) |
| IZOD Impact Resistance (Unnotched) | 449 J/m | ASTM D4812-11 (Unnotched) |
| Water Absorption | 0.54% | ASTM D570-98 (2018) |
Biocompatibility Certification
Samples printed with BioMed Clear Resin have been evaluated in accordance with ISO 10993-1: 2018 and ISO 7405: 2018, meeting the requirements for the following biological hazards:
| ISO Standard | Test Description3 |
| EN ISO 10993-5:2009 | Non-cytotoxic |
| ISO 10993-10:2010/(R)2014 | Non-irritating |
| ISO 10993-10:2010/(R)2014 | Non-sensitising |
| ISO 10993-3:2014 | Non-genotoxic |
| ISO 10993-11:2017 | Non-toxic |
ISO Standards Compliance
This product has been developed in strict compliance with the following standards:
| ISO Standard | Description |
| EN ISO 13485:2016 | Medical devices – Quality management systems – Requirements for regulatory purposes |
| EN ISO 14971:2012 | Medical devices – Application of risk management to medical devices |
1 Material properties may vary depending on part geometry, print orientation, Slicer settings, temperature, and the disinfection or sterilisation methods used.
2 Data measured on post-cured samples, printed on a Form 3B at 100-micron settings for BioMed Clear Resin, washed for 20 minutes in 99% isopropyl alcohol, and subsequently post-cured at 60°C for 60 minutes in a Form Cure.
3 BioMed Clear Resin was tested at NAMSA World, OH, USA.
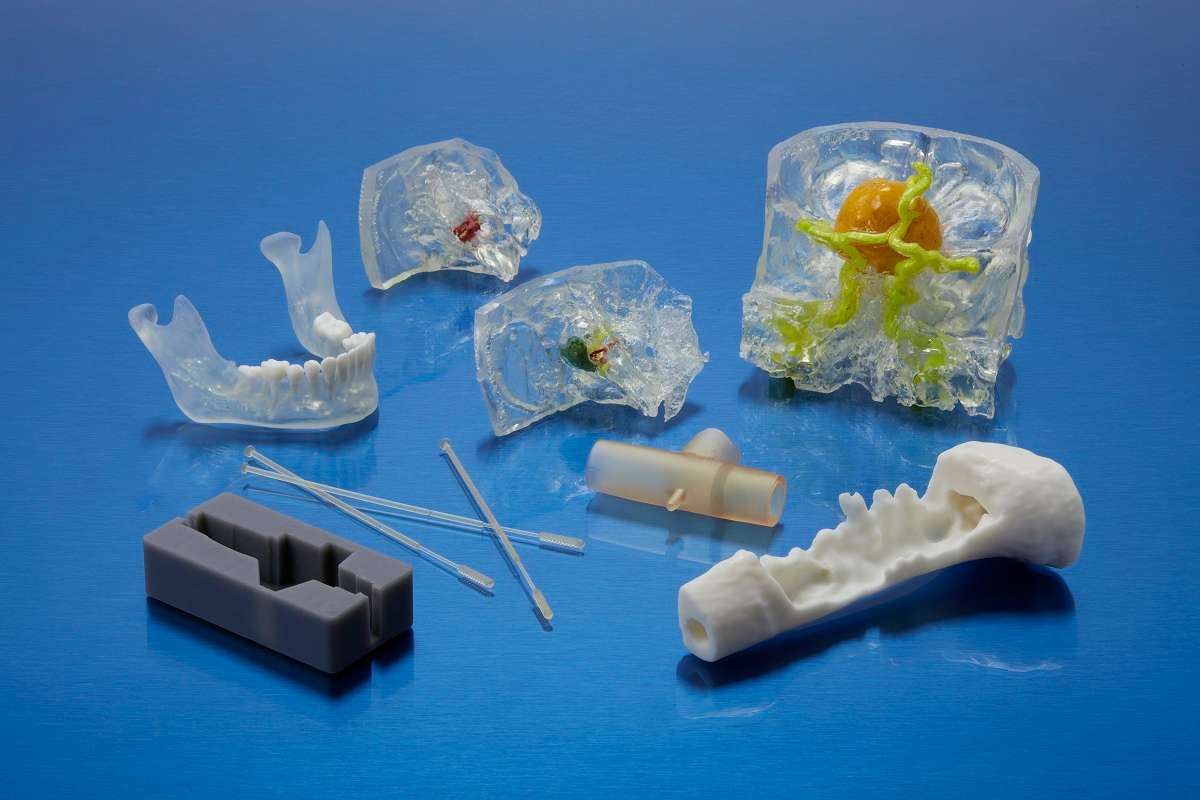
Video: 3D Printing Medical Devices with SLA Technology and BioMed Resin
Discover how to achieve professional, safe results with Formlabs printers:




























































