






- Stock: In Stock
- Product code: 00-00000267
- Weight Brutto: 1.50kg
- SKU: RS-F2-BMWH-01
Formlabs BioMed White Resin – Medical Resin
The Formlabs BioMed White Resin has been developed with the goal of raising the standard of healthcare. Thanks to medical 3D printing, you can now bring cutting-edge tools within reach of any healthcare centre or clinic.
Formlabs continuously expands its range of materials for its 3D printers. The Formlabs BioMed Resin line is designed for a wide variety of applications where performance and biocompatibility are critical. BioMed Resin materials are developed and manufactured in ISO 13485-certified facilities, and are fully compatible with standard disinfection and sterilisation methods.
Material for 3D Printing in the Medical Sector
BioMed White Resin is a rigid, opaque, and biocompatible photopolymer resin. It is designed for applications requiring prolonged skin contact or brief contact with patient mucous membranes. This unique medical resin has been tested for pyrogenicity and acute systemic toxicity in accordance with US Pharmacopeia standards. As such, it is ideal for cases involving short-duration contact with tissue, bone, and dentine.
Parts printed with BioMed White Resin can be processed using standard solvent disinfection and sterilisation methods. This material is produced in an ISO 13485-certified facility and holds USP Class VI certification, making it suitable for pharmaceutical and drug delivery applications.
Print resolution: 100 µm, 50 µm.
Requires final post-curing.
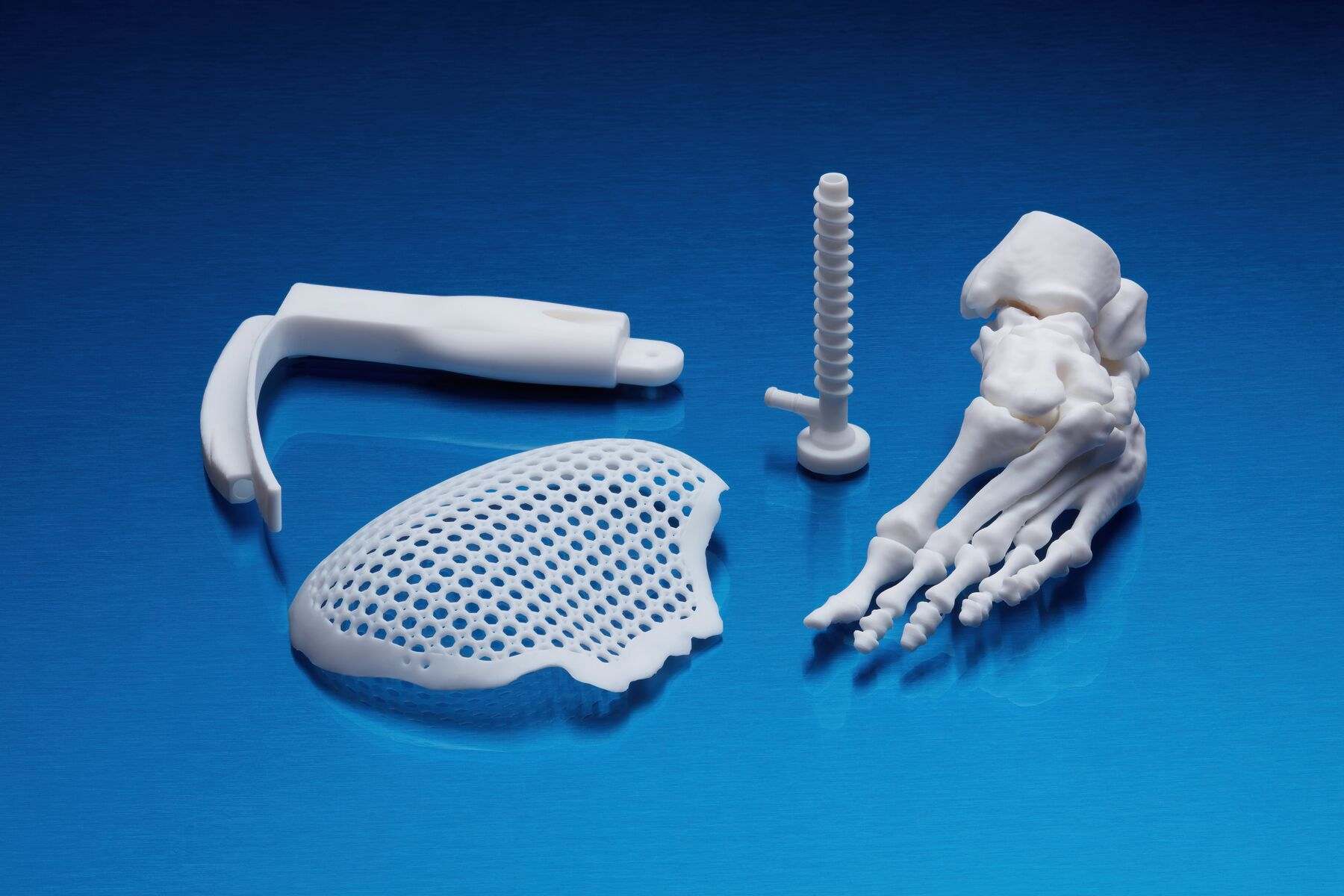
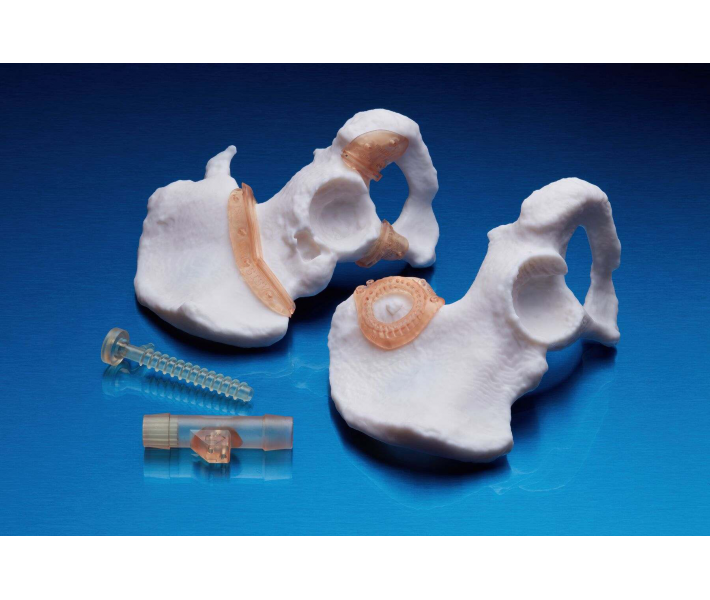
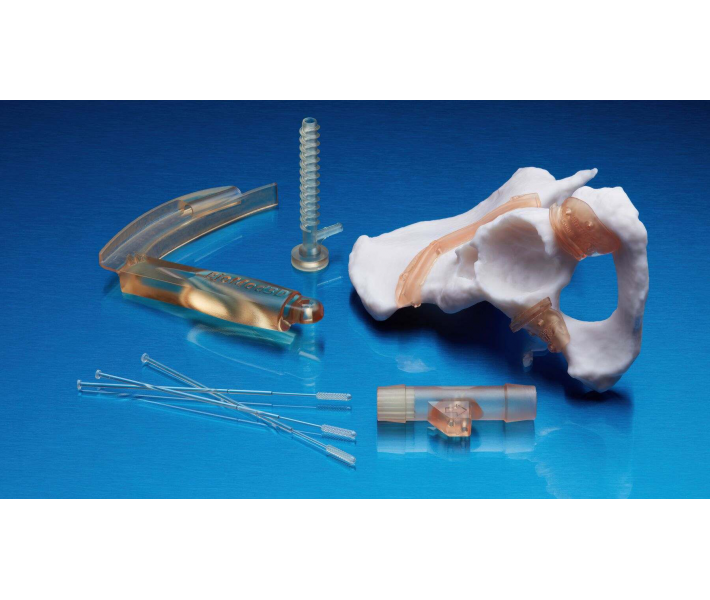
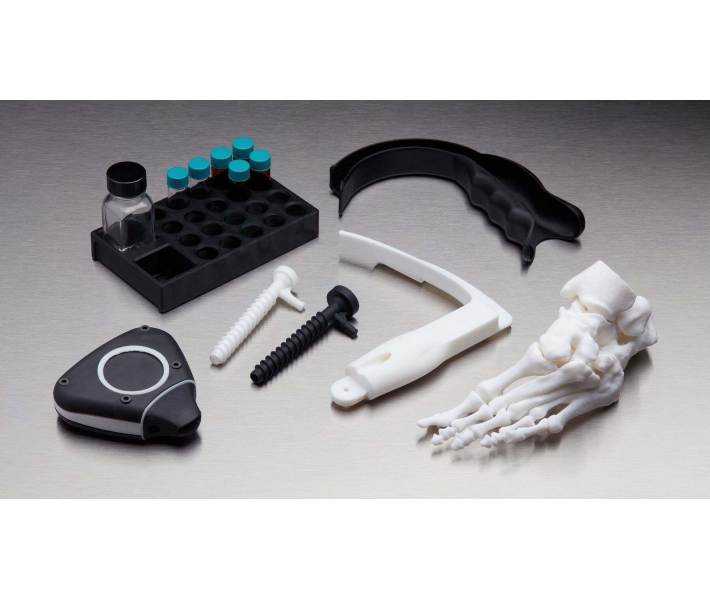
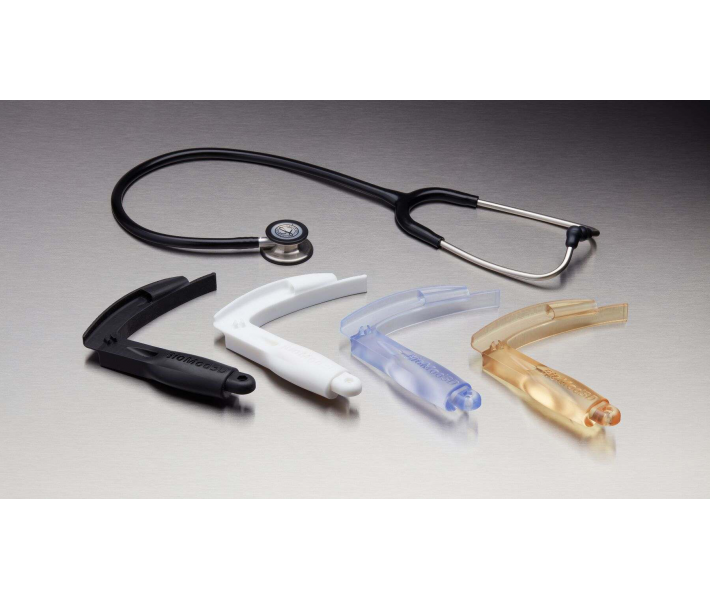
Applications of BioMed White Resin
This material enables rapid, customised production of:
- End-use medical devices and their components.
- Customised models and implant replicas for determining precise sizing.
- Cutting and drilling guides.
- Surgical guides and templates.
- Moulds, clamps, fixtures, and biocompatible devices.
- Anatomical models that can be used directly in the operating theatre.
Technical Properties of BioMed White Resin:
| After post-curing1,2 | Method | |
| Tensile Properties | ||
| Tensile Strength | 45.78 MPa | ASTM D 638-10 (Type IV) |
| Tensile Modulus | 2020.16 MPa | ASTM D 638-10 (Type IV) |
| Elongation | 10% | ASTM D 638-10 (Type IV) |
| Flexural Properties | ||
| Flexural Stress at 5% Strain | 74.46 MPa | ASTM D 790-15 (Method B) |
| Flexural Modulus | 2020.16 MPa | ASTM D 790-15 (Method B) |
| Hardness | ||
| Shore D Hardness | 80D | ASTM D 2240-15 (Type D) |
| Mechanical Properties | ||
| Izod Impact Resistance (notched) | 15.11 J/m | ASTM D 256-10 (Method A) |
| Izod Impact Resistance (unnotched) | 269.03 J/m | ASTM D 4812-11 |
| Thermal Properties | ||
| Heat Deflection Temperature at 1.8 MPa | 52.4 °C | ASTM D 648-18 (Method B) |
| Heat Deflection Temperature at 0.45 MPa | 67.0 °C | ASTM D 648-18 (Method B) |
| Coefficient of Thermal Expansion | 90.1 µm/m/°C | ASTM E 831-13 |
| Other Properties | ||
| Water Absorption | 0.40 % weight | ASTM D570-98 |
| Sterilisation Compatibility | |
| Electron Beam Radiation (E-beam) | Electron beam processing at a dose of 35 kGy |
| Ethylene Oxide | Immersion in 100% ethylene oxide solution at 55 °C for 180 minutes |
| Gamma Radiation | Gamma radiation processing at doses of 29.4 – 31.2 kGy |
| Steam Sterilisation |
Autoclave at 134 °C for 20 minutes Autoclave at 121 °C for 30 minutes |
| Disinfection Compatibility | |
| Chemical Disinfection | Immersion in 70% isopropyl alcohol solution for 5 minutes |
Biocompatible Medical Material
Samples printed with BioMed White Resin have been evaluated in accordance with the following biocompatibility standards:
| ISO Standard | Description3 |
| ISO 10993-5:2009 | Non-cytotoxic |
| ISO 10993-10:2010 | Non-irritating |
| ISO 10993-10:2010 | Non-sensitising |
| ISO 10993-11: 2017 | No signs of acute systemic toxicity |
| ISO 10993-11: 2017 | Non-pyrogenic |
The product has been developed in compliance with the following ISO standards:
| ISO Standard | Description |
| EN ISO 13485:2016 | Medical devices – Quality management systems – Requirements for regulatory purposes |
| EN ISO 14971:2012 | Medical devices – Application of risk management to medical devices |
Solvent Compatibility of BioMed White Resin
Percentage weight increase after 24 hours for a printed cube of 1 x 1 x 1 cm after final post-curing, immersed in the corresponding solvent:
| Solvent | Weight gain after 24h, % | Solvent | Weight gain after 24h, % |
| Acetic acid, 5% | 0.4 | Mineral oil, heavy | < 0.1 |
| Acetone | 2.9 | Mineral oil, light | < 0.1 |
| Bleach, ~5% NaOCl | 0.3 | Salt water (3.5% NaCl) | 0.4 |
| Butyl acetate | 0.4 | Skydrol 5 | 0.5 |
| Diesel fuel | < 0.1 | Sodium hydroxide (0.025%, pH = 10) | 0.3 |
| Diethylene glycol monomethyl ether | 1.0 | Strong acid (conc. HCl) | 0.2 |
| Hydraulic oil | < 0.1 | Tripropylene glycol monomethyl ether | 0.6 |
| Hydrogen peroxide (3%) | 0.3 | Water | 0.3 |
| Isooctane | < 0.1 | Xylene | 0.3 |
| Isopropyl alcohol | 0.2 |
































































